Lardone R. et al. J Biomed Sci. 2019
BACKGROUND: Different neurological disorders frequently display antibodies against several self-glycans. Increasing evidence supports their pathogenic role; however, far less is known about their origin. Meanwhile, antibodies recognizing non-self glycans appear in normal human serum during immune response to bacteria.METHODS: Using high performance thin layer chromatography-immunostaining, we comparatively evaluated humoral immune response (IgG and IgM immunoreactivity) against glycolipids carrying self-glycans (GM3/GM2/GM1/GD1a/GD1b/GD3/GT1b/GQ1b) and non-self glycans (Forssman/GA1/”A” blood group/Nt7) in sera from 383 patients with neurological disorders along with 87 healthy controls.
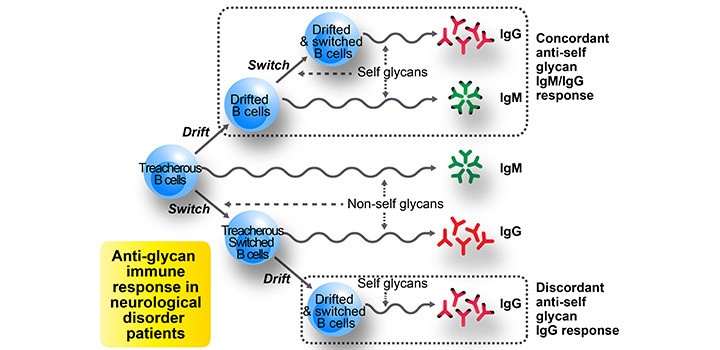
RESULTS: In contrast to no healthy controls having anti-self glycan IgG antibodies, one-fifth of patients’ sera had anti-self glycan IgG antibodies: remarkably, 60% of these occurred without IgM antibodies of the same specificity. Contrary to this unusual fact (anti-self glycan IgG occurrence without simultaneous presence of IgM having the same specificity ~ IgG/IgM discordance), all IgG antibodies against non-self glycans occurred simultaneously with their IgM antibody counterpart (i.e. 0% discordance). When analyzed closer, the IgG/IgM discordance frequency for anti-self glycans exhibited a dual trend: below 40% for IgG antibodies against GM2, GM1 and GD1b, and greater than 53% for IgG antibodies against the remaining self glycans. Interestingly, this discordance behavior was common to several different neurological disorders.
CONCLUSIONS: Classic immunology principles indicate this anti-self glycan IgG/IgM discordance should not occur in an antibody response; its unusual presence is discussed within the “binding site drift hypothesis” context, where anti-self glycan IgG antibodies could originate from pre-existing IgG recognizing structurally-related non-self glycans..
Authors: Lardone RD, Irazoqui FJ, Nores GA