Gallea JI, et al. JNC. 2018
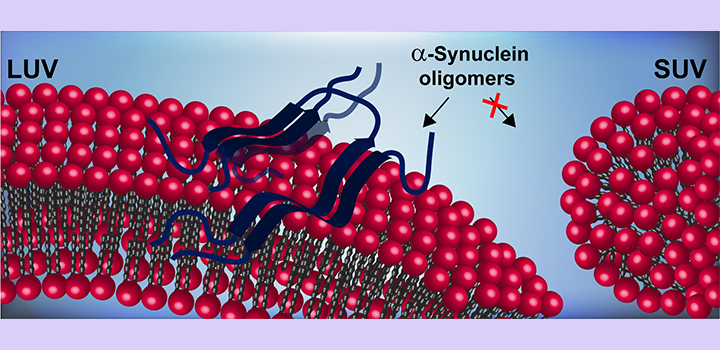
The amyloid aggregation of the presynaptic protein α-synuclein (AS) is pathognomonic of Parkinson’s disease and other neurodegenerative disorders. Physiologically, AS contributes to synaptic homeostasis by participating in vesicle maintenance, trafficking and release. Its avidity for highly curved acidic membranes has been related to the distinct chemistry of the N-terminal amphipathic helix adopted upon binding to appropriated lipid interfaces. Pathologically, AS populate a myriad of toxic aggregates ranging from soluble oligomers to insoluble amyloid fibrils. Different gain-of-toxic function mechanisms are linked to prefibrillar oligomers which are considered as the most neurotoxic species. Here, we investigated if amyloid oligomerization could hamper AS function as a membrane curvature sensor. We used fluorescence correlation spectroscopy to quantitatively evaluate the interaction of oligomeric species, produced using a popular method based on lyophilization and rehydration, to lipid vesicles of different curvatures and compositions. We found that AS oligomerization has a profound impact on protein-lipid interaction, altering binding affinity and/or curvature sensitivity depending on membrane composition. Our work provides novel insights into how the formation of prefibrillar intermediate species could contribute to neurodegeneration due to a loss-of-function mechanism.
Autores: Gallea JI, Ambroggio EE, Vilcaes AA, James NG, Jameson DM, Celej MS.